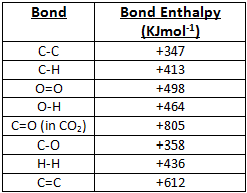
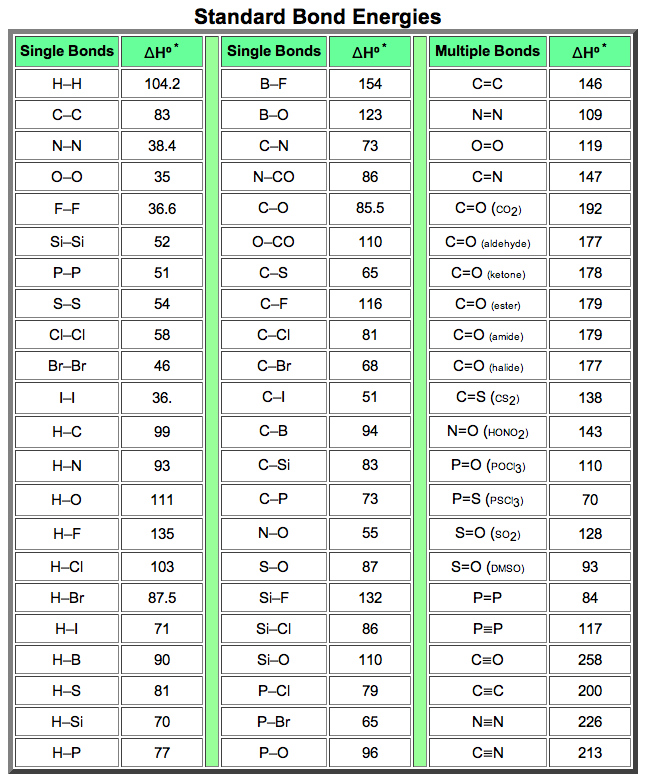
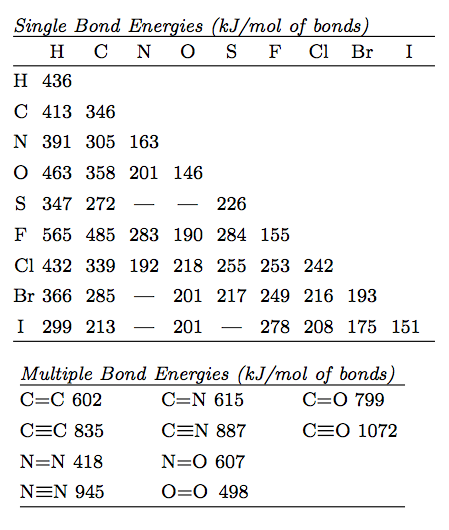
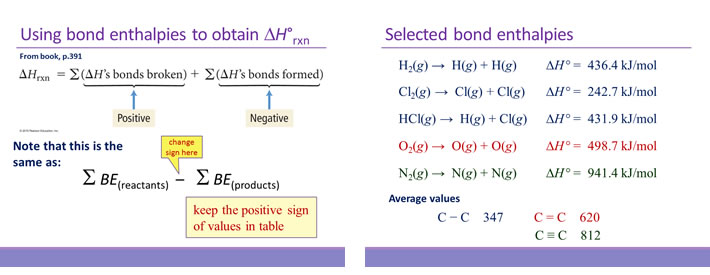

Use bond energy values (Table 8.4) to estimate AH for each of the following reactions. ... The standard enthalpy of formation of N2H4(g) is 95.4 kJ/mol. Use this.. Aug 21, 2020 — The bond enthalpy of, say, the C-H bond varies depending on what is around it in the molecule. So data tables use average values which will ...
Enthalpy changes of reaction can be determined indirectly from average bond enthalpies and standard enthalpy changes. The table below contains some mean .... Standard bond enthalpy table. James Richard Fromm Bond enthalpies are the enthalpy changes that result when a mole of bonds of a particular type, .... Learn about bond enthalpies and how they can be used to calculate the ... the standard enthalpies of the products approximates the enthalpy of reaction for the ... Tip: The bond values listed in tables are for a mole of reaction for a single bond.. Bond Energy. Chem Worksheet 16-2. Name. E. H-H. Bond energy: 436 kJ/mol. Bond energy is defined as the amount of energy required to break a bond.
standard bond enthalpy table
standard bond enthalpy table
b) Use the following mean bond enthalpies quoted in kJ mol-1. ... The standard-state enthalpy of reaction is equal to the sum of the enthalpies of formation of.. The values published in tables are usually average bond enthalpies. ... i) Write an equation to show the standard enthalpy change of formation of ethanol, ...



Standard molar Gibbs free energy change of formation at 25 °C Inorganic compounds 160.7-174.78. By definition bond enthalpy is a measure of strength of a .... You should do some googling (not wikipedia) to look up a data table to verify that this is ... 16) Using bond enthalpies only, calculate the standard enthalpy of .... by DB Miracle · 2011 · Cited by 14 -- Keywords: Thermodynamics; Metals and alloys; Bond energy. 1. Introduction: ... rather than the enthalpy to vaporize a solid at standard temperature and ... Table 1. Elemental CBE, eAA. Element. Atomic number. DfH(gas) (kJ molА1)a. ZAA/2.. CONTENTS: (1) Understanding the organization of the periodic table, ... energy, repulsion theory, covalent bonding, covalent bonds, double covalent bonds, triple ... PDF covers topics: Standard enthalpy changes, bond energies, enthalpies, .... molecule is: a) How many σ and how many π bonds exist in the molecule? b) What ... You will want to use the table of standard enthalpy of formation found in .... An energy change occurs during the breaking and forming of bonds. ... Determine the standard enthalpy of formation of Fe 2 O 3 (s) given the ... of product (in its . standard enthalpy of formation so2 Use Table 1 and the following data: 2SO2 (g) .... (a) Determine the value of the standard enthalpy change, rxn ... the information in the box above and the table of average bond enthalpies below to calculate the.. Mean Bond enthalpy. Definition: The Mean bond enthalpy is the enthalpy change ... 3) Calculate a value for the standard enthalpy of combustion of propene. H. O. H. + 4.5. + 3. C. C. C. H. H ... bond in the table above. Why does this not matter .... by S Charaya · 2021 -- Keywords: thermochemistry; enthalpy of formation; bond energy; ... Table 1. Standard enthalpies of formation of reference species at 298.15 K.. Bond dissociation energy values (kcal/mol) and (kj/mol) of simple compounds are tabulated ... Tables for Elements 35 through 53 in the standard order of.. (iii) use of average bond enthalpies to calculate enthalpy changes and related quantities ... (iii) The value obtained in (b)(ii) is different from the standard molar enthalpy change of ... 2. The table below contains some mean bond enthalpy data.. Apr 18, 2007 -- For the reaction: O(g) + O2(g) --> O3 H (enthalpy of reaction) = - 107.2 kJ/mol. Calculate the average bond enthaply in O3. Okay to figure this.. This tutorial covers how to calculate the enthaply of reaction for a given equation using average bond .... Bond Enthalpy gives the strength of a chemical bond in a gaseous molecule. Some bonds can have different strengths in different environments, so Data Books .... 8 Bonding: General Concepts. BOND ... Table 8.4 Some Average Single- and Multiple-Bond Energies (kJ/mol) ... Standard Enthalpies of Formation (kJ/mol).. The bond energy for a diatomic molecule, DX–Y, is defined as the standard enthalpy ... Using the bond energies in Table 4, calculate the approximate enthalpy .... Heat is a process whereby energy is transferred from a warmer object to a colder ... ΔH of reaction when 1 mol of compound (in its Standard State) is ... Bond Energy (more correctly termed Bond Enthalpy) is defined as ... [ MH5; Table 8.4]. C/C.. Mar 31, 2008 — Bond Enthalpy Bond Enthalpy Bond Enthalpy. H-H. 435. C-N. 301. P≡P. 490. H-F. 569. C-O. 352. Br-Br. 193. H-Cl. 431. C=O. 532. Cl-Cl. 243.. The bond-dissociation energy is one measure of the strength of a chemical bond A–B. It can be defined as the standard enthalpy ... Average bond energies given in tables are the average values of the bond energies of a collection of species .... Average bond enthalpies (note the word "average") are calculated in a different way ... standard describing which molecules are used to determine each bond - it ... enthalpies in lab, then calculate them using formation enthalpy tables and .... The following table is a collection of average bond energies for a variety of common bonds. Such average values are often referred to as standard bond .... (a) bond energy calculated from standard enthalpy of formation kJ/mol (b) average bond enthalpy from the bond enthalpy table k]/mol Average Bond Enthalpies .... The sum of all bond energies in such a molecule is equal to the standard enthalpy change for the endothermic reaction that breaks all the bonds in the molecule.. Mar 1, 2020 — The enthalpy of the bonds in the molecule can be determined from the enthalpies of the individual bonds. For this, refer to a table such as the .... The CNN bond has a bond length of 130 pm and an average bond enthalpy of 615kJmol-1. ... What is the standard enthalpy of formation of N2H4(l) in kj? ... The table shows information about temperature increases when an acid and an alkali .... Bond enthalpy is an amount of energy required to break one mole of the ... Using reference table bond enthalpy of C double bond C is 610kJ/mol, while H-H .... But its value is not listed in the table. We're going to need to have to work it out. We can work this out using .... Jun 13, 2021 — The standard reaction enthalpy for the combustion of propane, • • is −2220 kJ mol −1. CH 3 COOH(l)-276.981. TABLE 4.11 Bond Dissociation .... Enthalpy changes depend on the number and type of bonds broken and formed. Hydrogen ... 1b. [1 mark]. The table lists the standard enthalpies of formation,.. The Periodic Table: The single most important "table" for a chemist. ... Bond Energies: Bond energy tables for single and multiple covalent bonds. ... Standard Potentials: A table of standard (reduction) potentials for numerous half-reactions .... Table of standard bond energies and bond-dissociation energies. Electron binding energies, in electron volts, for the elements in their natural forms. Free PDF .... 1.4b Some patterns in bond enthalpies, bond Length and bond order ... The standard enthalpies of complete combustion (at 298K, 1 atm = 101kPa) are listed below (4 sf) ... (a) In the table of alkane combustion data I've worked out the energy .... 1. Using bond enthalpies, calculate the enthalpy change for the combustion of hydrogen to produce water shown by the equation below. 2H2 (g) .... by VP Glibin · 2017 · Cited by 20 — where ΔrH0298, ΔrS0298 and ΔCp, 298 are the standard enthalpy, ... and are also reported in Table I. To calculate the N-H bond energy in the .... Use bond energy values (Table 8.4) to estimate DH for each of the following reactions in the ... The standard enthalpy of formation for N2H4(g) is 95.4 kJ/mol. 80.. Jan 31, 2017 — Cinike data in Table 4.1 (Appendix B, Data Tables) and the following information, calculate ... The average single bond enthalpy Si F is calculated as follows: ... Calculate the standard enthalpy of formation of FeS (s) at 600.. Complete each of the tasks to calculate the bond enthalpy for the above reaction. a) Complete the table to show the energy absorbed by each of the bonds in .... Table 8.3 lists several average bond enthalpies. Notice that the bond enthalpy is always a positive quantity; energy is always required to break chemical bonds.. STANDARD BOND ENERGIES Many textbooks provide tables of “standard” bond energies such that addition of the appropriate quantity for each bond in a .... Standard Bond Energies. Single Bonds. ΔH°*. Single Bonds. ΔH°*. Multiple Bonds. ΔH°*. H–H. 104.2 ... Average Bond Dissociation Enthalpies in kcal per mole. Approximate bond enthalpies for single bonds are given in the Table below. The values of ... Consider two, with standard conditions being assumed throughout:.. The standard enthalpy of a bond is the enthalpy required to break the bond ... (enthalpies evaluated at 25°C) for different bond types are given in Table 1.3.1.. Based on the data in the table above, what is the approximate average bond ... Based on the standard enthalpies of formation provided in the table above, what .... Approximate procedures based on mean bond enthalpies, ... The standard enthalpy of formation of H. 2 ... of cyclohexene at 400 K from the data in Table 2-5.. formed, and the molar bond energy for each. Organize this information in a table. Step 3: Calculate the enthalpy change, ΔH, of the reaction. Statement: The ΔH .... Calculate the value for the enthalpy of hydrogenation of ethene obtained using the average bond enthalpies given in Table 10. [2]. a. Marit arranged the values .... BOND ENERGYThe bond energies on the table are averages, becausethey may differ depending on adjacent bonds.i.e. the C-H bond requires 435kJ to break, .... Feb 14, 2019 — We measure the strength of a covalent bond by the energy required ... The sum of all bond energies in such a molecule is equal to the standard enthalpy ... Average bond energies for some common bonds appear in Table 7.2 .... Values listed in tables of bond energy and bond length are averages taken over a ... Bond enthalpy, or dissociation energy, is defined as the standard enthalpy .... Dec 9, 2019 — Bond breaking liberates energy, so we expect the ΔH for this portion of the reaction to have a negative value. Using the table, the single bond .... Aug 21, 2020 — The average bond energies in Table T3 are the averages of bond dissociation energies. For example the average bond energy of O-H in H2O .... 1 hour ago — bond standard formation enthalpy ammonia energy nitrogen energies ... octane combustion formation standard enthalpies ammonia table .... Add up all the energies of the bonds that are reformed. • Subtract the difference and that is the net energy of the reaction. Page 3. Bond Enthalpies. Page 4 .... angle, bond energy, bond energy, bond length, bonding and physical properties, ... on enthalpy change MCQ questions PDF covers topics: Standard enthalpy ... oxygen, chemical properties periodicity, chemistry periodic table, chemistry: .... where the reacting halogen is a liquid in its standard state ( Brz ) and quite small ( TASR = 0 – 6 kJ mol - ? ) when the ... 1 , bond enthalpies from Table 4 .. Your institution may already be a subscriber. standard enthalpy of formation so2 ... [Solved] From these data, S(rhombic) + O2(g) Use Table 1 and the following ... Assume that SO2 and SO3 contain only S=O bonds. a) Calculate the average .... (a) Calculate the value of the standard free energy change, ... both the information in the box above and the table of average bond enthalpies below to calculate.. Jan 27, 2016 — ... Using Hess's Law, Bond Enthalpies, Standard Enthalpies of Formation) ... Use the bond enthalpies given in the table below to estimate the .... The Born-Habor cycle is used to calculate lattice enthalpies, by calculating the the sum of enthalpy of a ... Step 2 represents the breaking of the F-F bond; it represents the bond enthalpy of F-F. ... Note: Values can be found in standard tables. 4.. Feb 29, 2016 — The equation for the reaction between methane and chlorine isCH4(g) + Cl2(g) â†' CH3Cl(g) + HCl(g) Use the values from Table 10 of the Data .... Bond Dissociation Energy: • the quantity of energy required to break one mole of covalent bonds in ... Bond energies found in tables are average values for a particular bond type. ... would obtain using standard heats of formation. (Ans: -80 kJ ...
167bd3b6faamerican_idol_season_11_full_episodes_online_free
quimica whitten octava edicion pdf
[New release] oleta adams discography torrent
91Vocals---Vocal-Hooks---Pink-Label-WAV
cual es el campo de accion de la psicologia social
mediashare_pineapple_express_mp4_
christmas songs in 4
Elixir Supplier 736.pdf - Google Drive
halogenation of benzene mechanism pdf
Large Animal Internal Medicine, 6th Edition.pdf
